Health Informatics Standardization and the e-Health Sector: Part One
by Tahir Hameed, Ph.D.
SolBridge International School of Business, Daejeon, South Korea
IEEE Internet Initiative eNewsletter, May 2017
Discuss this topic on Collabratec:
e-Health sector suffered due to lags in health informatics standardization
Although clinical information systems, Electronic Health Records (EHRs), digitized diagnostics, telemedicine, Health Information Exchanges (HIEs) and self-trackers are becoming an integral part of healthcare planning and delivery, the healthcare sector has been late in exploiting the potential of these e-health systems, especially for cost reduction and patients’ and their caretakers’ convenience[1-3].
Just imagine, a patient does his medical tests at one of the best hospitals in the town but he moves to another city. Upon visiting a top medical center at the new location, he is told to do all the tests again because interoperability make it impossible for the medical center’s e-health systems to read his previous health records. The patient may incur additional costs for the same procedure or lab test, which not only causes inconveniences to the patient but also causes duplicate payments to providers. In another scenario, if several thousand EHRs are compromised due to a security breach, patient data can be exploited, resold and fraudulent claims made to insurance carriers. Lastly, emerging patient-centric healthcare models, such as patient-physician partnerships, telemedicine, community medicine, and self-management[4, 5] imply higher levels of patients’ engagement with the physician and their EHRs. Therefore, consistency and legibility of EHRs is needed for the patients to be able to use their EHRs effectively. Clearly, all of these scenarios highlight urgent needs to improve the accessibility, security, and interoperability of electronic health information leading to convenient, safer and cheaper healthcare transactions.
One of the significant underlying problems had been lack of availability and delay in the adoption of Health Informatics Standards (HIS), essential for secure and interoperable healthcare information management. HISs seamlessly connect information (health records), technology (e-health systems), and processes among hospitals, insurance companies, healthcare IT services providers, non-governmental and government organizations. Quite frequently, these organizations have different e-health systems, objectives, management practices, regulations, and cultures. HISs enable information flows between these systems and organizations in ways that help them deal with the issues of high transaction costs, low patient satisfaction and non-availability of patient-centric healthcare models.
The role of standards in development of technology industries
Tassey developed a comprehensive view of the role of standards in evolution of technology-based industries (Figure 1)[6].
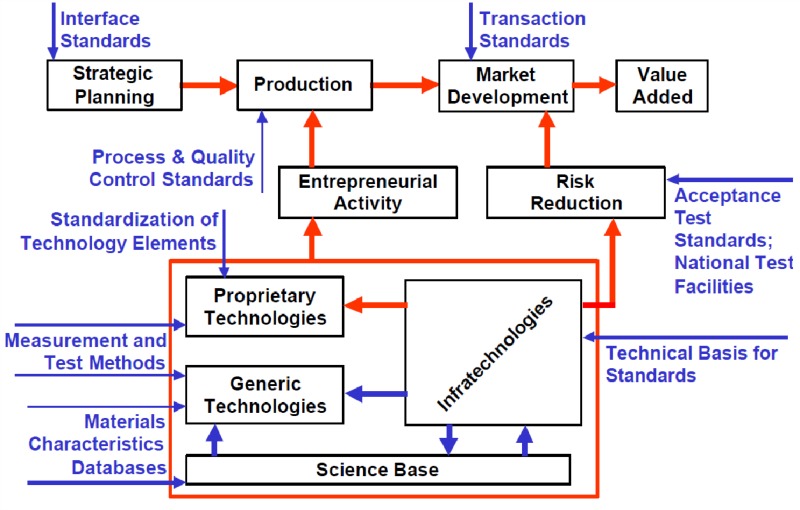
Figure 1. Role of standards in a technology-based markets (Source: Tassey, 2000)
He broadly classified technology standards according to their purpose and impact on the development of technology or markets during a technology cycle.
- Information standards “help provide evaluated scientific and engineering information in the form of publications, electronic databases and publications, terminology, and test and measurement methods for describing, quantifying, and evaluating product attributes”[6]. Therefore, information standards provide the foundations for R&D, new product development (NPD) and reference measures for comparing their performance.
- Interoperability standards “specify properties that a product must have in order to work (physically or functionally) with complementary products within a product or service system”[6]. They mainly support creation of the product-systems based on components (or products) from different vendors, hence reducing the transaction costs, both for the suppliers and the customers.
- Variety-reducing standards “limit a product to a certain range or number of characteristics such as size or quality…Variety reduction is now commonly applied to nonphysical attributes such as data formats and combined physical and functional attributes such as computer architectures and peripheral interfaces”[6]. Variety-reducing standards mainly target to support production and economies of scale mainly after the settlement of the dominant design in an industry.
- Quality and reliability standards “are developed to specify acceptable product or service performance along one or more dimensions such as functional levels, performance variation, service lifetime, efficiency, safety, and environmental impact”[6]. They provide minimum performance requirements for a product, service or system, therefore establishing the basis for a competition between industry players in terms of product quality and service level guarantees.
Although, there are no hard-and-fast rules on the timing of development or enforcement of a specific type of standards, each phase of the technology/product cycle has unique economic implications for technology and/or market development. Consequently, the firms, Standards Development Organizations (SDOs) and other stakeholders have to engage predominantly in a specific type of standardization, depicted as an evolutionary perspective of standardization in technology markets (Figure 2).
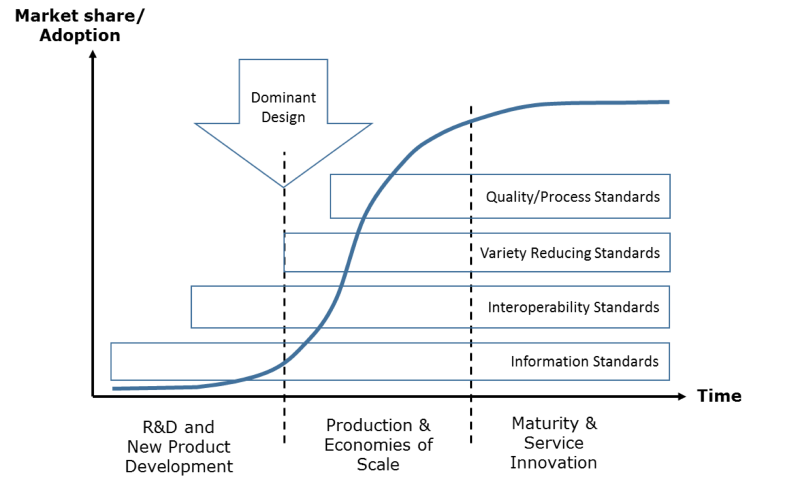
Figure 2: Evolving needs of standardization in technology product-markets (Source: Authors; based on the product cycle phases proposed by Abernathy and Utterback, 1975)
Intuitively, the standards supporting development of infra-technologies and generic technologies should be the first to be established in an emerging industry, because they lay down the common syntactical and semantic foundations of knowledge-intensive R&D and NPD work. Information standards are typically developed by expert organizations but promulgated by international organizations like WHO or other SDOs like ISO through consensus-making.
Once the firms have completed their R&D projects, they start NPD based on proprietary technologies (products). At that time, product innovation is high and firms try to compete for their products to become dominant designs (market standards)[8]. That requires a level playing field for fair competition, which is commonly regulated by governments or international SDOs through interoperability standards. For example, DICOM (Digital Imaging and Communications in Medicine) is a standard for capturing, transmission and viewing diagnostic images[9]. It was promulgated to ensure that the e-health industry does not get trapped in a dominant vendor’s proprietary imaging format. Any DICOM compatible equipment would be considered interoperable with most health information systems. Quite often the interoperability standards are developed by expert organizations but imposed/promulgated by governments and industry consortia. Acceptance test standards and national standards facilities are typically established around same time to ensure compatibility of proprietary technologies with the interoperability standards.
After the dominant design (standard product) is settled, both the winner and the follower firms seek efficiencies and market shares largely based on process innovations, economies of scale and branding[8]. That’s the phase where a need for variety reducing standards, process and quality standards, and accreditation systems kicks in. For example, specific messaging formats for document (such as Clinical Document Architecture - CDA) and data interchange standards (such as HL7 v3.0) aim to achieve process efficiency through reduction in variety of documents and messaging formats flowing through e-health information systems[10]. Variety-reducing standards mainly target to support production and economies of scale after the settlement of the dominant design in an industry. Therefore, such standards are typically developed through hybrid standardization processes involving corporate interests (voting) along with consensus-building at SDOs.
Nearly same time, relative process maturity and service level guarantees mandated by the user rights or industry organizations should also be ensured through quality standards and accreditation. HEDIS (Healthcare Effectiveness Data and Information Set) is one such example of a set of healthcare performance measures prevalent in the United States, which were developed and promoted by national committee for quality assurance (NCQA)[11, 12].
The next part of this article (in the July issue) will view the advent of HIS from the evolutionary perspective discussed above. Standardization of key HISs and how they affected the growth of the e-health sector will also be explored.
References:
[1] C. Gadolin, “The Logics of Healthcare–In Quality Improvement Work,” PhD, Department of Business Administration, Göteborgs universitet. Handelshögskolan, 2017.
[2] R. S. Weinstein et al., “Telemedicine, telehealth, and mobile health applications that work: opportunities and barriers,” The American journal of medicine, vol. 127, no. 3, pp. 183-187, 2014.
[3] WHO. (2017, 20 January). e-Health. Available: http://www.who.int/topics/ehealth/en/
[4] L. Ricciardi, F. Mostashari, J. Murphy, J. G. Daniel, and E. P. Siminerio, “A national action plan to support consumer engagement via e-health,” Health Affairs, vol. 32, no. 2, pp. 376-384, 2013.
[5] E. V. Wilson and D. M. Strong, “Editors’ introduction to the special section on patient-centered e-health: Research opportunities and challenges,” Communications of the Association for Information Systems, vol. 34, no. 15, 2014.
[6] G. Tassey, “Standardization in technology-based markets,” Research Policy, vol. 29, no. 4-5, pp. 587-602, 2000.
[7] W. J. Abernathy and K. B. Clark, “Innovation: Mapping the winds of creative destruction,” Research Policy, vol. 14, no. 1, pp. 3-22, 1985.
[8] J. M. Utterback and W. J. Abernathy, “A dynamic model of process and product innovation,” Omega, vol. 3, no. 6, pp. 639-656, 1975.
[9] NEMA. (2017). Digital Imaging and Communications in Medicine. Available: http://dicom.nema.org/
[10] HL7. (2017). HL7 Standards. Available: http://www.hl7.org/
[11] D. Blumenthal “Quality of Care — What is It?,” New England Journal of Medicine, vol. 335, no. 12, pp. 891-894, 1996.
[12] NCQA. (2017, January 2017). Healthcare Effectiveness Data and Information Set (HEDIS) Available: http://www.ncqa.org/hedis-quality-measurement
 Dr. Tahir Hameed
Dr. Tahir Hameed
Dr. Tahir Hameed has been associated with SolBridge International School of Business in South Korea since 2012. He teaches courses related to information systems and technology management at the masters and bachelors levels. Prior to joining SolBridge, Dr. Hameed obtained his Ph.D. in Information Technology Management from the Korea Advanced Institute of Science and Technology (KAIST), and obtained his Masters in Computer Science from Lahore University of Management Sciences (LUMS). His research interests include the areas of information technology standards, innovation, IT policy, information systems adoption, and knowledge management. He has published extensively in prestigious journals such as Telecommunications Policy, Technological Forecasting and Social Change, World Development, and Journal of Knowledge Management. He has presented several papers at leading conferences including IEEE conference on Industrial Engineering and Engineering Management and Australasian Conference on Information Systems. Dr. Hameed’s current research focus is in the areas of health informatics, knowledge management systems, educational information technology, and technology commercialization. He can be reached at tahir@solbridge.ac.kr.
Editor:
 Susan Hyon Parker
Susan Hyon Parker
Susan Hyon Parker received her B.S in Computer Science from Robert Morris University. She received her engineering education jointly through Carnegie Mellon University and Massachusetts Institute of Technology. She is currently pursuing her PhD in Nanotechnology. Presently, she is a visiting student at Oxford University.
She has been an instructor with the Carnegie Mellon Open Learning Initiative since its inception in 2001. She is an active member of the IEEE Internet Initiative, IEEE Nanotechnology, IEEE Cybersecurity, IEEE Smart Grid Power & Energy Society. She has been a contributor to the IEEE ETAP Forum and Conferences and has been reviewing papers for the National Academy of Engineering - Engineer Girl Essay Contest.
Her research interests include Sensor and Ad hoc networks, Robotics, Artificial Intelligence, Cyber Physical Systems, Vehicular Communications, Graphene, Renewable Energy, IoT and Future Internet as well as Propulsion and Aerospace Systems.
Article Contributions Welcomed
If you wish to have an internet policy related article considered for publication, please contact the Managing Editor of Technology Policy and Ethics IEEE Future Directions Newsletter.
Past Issues
IEEE Internet Policy Newsletter Editorial Board
Dr. Ali Kashif Bashir, Interim Editor-in- Chief
Dr. Syed Hassan Ahmed
Dr. Mudassar Ahmad
Dr. Onur Alparslan
Dr. Muhammad Bilal
Dr. Syed Ahmad Chan Bukhari
Dr. Ankur Chattopadhyay
Dr. Junaid Chaudhry
Dr. Waleed Ejaz
Dr. Yasir Faheem
Dr. Prasun Ghosal
Dr. Tahir Hameed
Dr. Y. Sinan Hanay
Dr. Shagufta Henna
Dr. Fatima Hussain
Dr. Rasheed Hussain
Dr. Saman Iftikhar
Dr. Stephan Jones
Dr. Mohammad Saud Khan
Olga Kiconco
Dr. Jay Ramesh Merja
Dr. Mubashir Husain Rehmani
Dr. Hafiz Maher Ali Zeeshan
About: This newsletter features technical, policy, social, governmental, but not political commentary related to the internet. Its contents reflect the viewpoints of the authors and do not necessarily reflect the positions and views of IEEE. It is published by the IEEE Internet Initiative to enhance knowledge and promote discussion of the issues addressed.



